A Vertically Integrated Medical Cannabis Company
Cultivation, Pharmaceutical Processing, and Quality Control
The Science of Precision: Pharmaceutical Cannabis Production at Anthera
At Anthera Pharmaceuticals, production begins in tightly controlled greenhouse environments where critical parameters are continuously monitored to support the cultivation of pharmaceutical-grade cannabis under GACP-compliant practices. Environmental conditions, plant development, and cultivation inputs are managed according to defined procedures, ensuring uniformity, traceability, and consistent plant quality. This disciplined approach establishes a robust foundation for both dried flower production and subsequent pharmaceutical manufacturing activities.
Following harvest, plant material is transferred within our integrated facility to EU-GMP-compliant processing areas, where validated procedures govern drying, trimming, sorting, and packaging of dried flower, as well as extraction and formulation of pharmaceutical products. Each stage is performed under controlled conditions with in-process monitoring, in-house quality control testing, and full batch traceability. This vertically integrated model ensures regulatory compliance, reproducibility, and consistent product quality from cultivation through to finished pharmaceutical release.
Manufacturing process
Manufacturing process
- Cultivation
- Post-Harvest Processing
- PACKAGING
- EU-GMP Quality Control Testing
- Research & Development
- Cultivation
- Post-Harvest Processing
- Extraction
- Packaging
- EU-GMP quality Control testing
- Research & Development





Cultivation
At Anthera Pharmaceuticals, cultivation is conducted in accordance with Good Agricultural and Collection Practice (GACP) to ensure consistent, traceable, and high-quality plant material for pharmaceutical use. Plants are propagated from selected genetics and grown under controlled greenhouse conditions using standardized procedures and documented workflows.
Key environmental parameters, including light, temperature, humidity, irrigation, and nutrition, are continuously monitored and adjusted according to cultivar-specific requirements. Crop development is managed to ensure uniformity, plant health, and reproducible outcomes. Harvest is performed based on predefined maturity criteria and quality indicators, ensuring that each batch of plant material meets the requirements for downstream pharmaceutical manufacturing.




Post-Harvest Processing
Immediately after harvest, plant material enters Anthera’s EU-GMP manufacturing areas, where all subsequent processing steps are performed under Good Manufacturing Practice. From this stage onward, the material is handled as a pharmaceutical starting material within a fully controlled, documented, and validated production environment.
Post-harvest manufacturing includes drying, trimming, and curing, each executed according to approved manufacturing instructions and under defined environmental conditions. These steps are designed to achieve controlled moisture reduction, physical standardization of the flower, and stabilization of the finished product while preserving critical quality attributes. All operations are monitored, recorded, and reviewed to ensure batch-to-batch consistency and full regulatory compliance.
Packaging
Packaging is performed under EU-GMP conditions using validated equipment under a controlled environment to maintain product quality and stability. Our packaging system is designed to minimize exposure to light, oxygen, and moisture, to prevent contamination and maintain product freshness and stability over the shelf life.
Each step in our packaging operation is subject to validated in-process controls with detailed documentation. Finished packages are labeled and secured in accordance with strict regulatory requirements, including complete traceability from cultivation batch to final medical product.




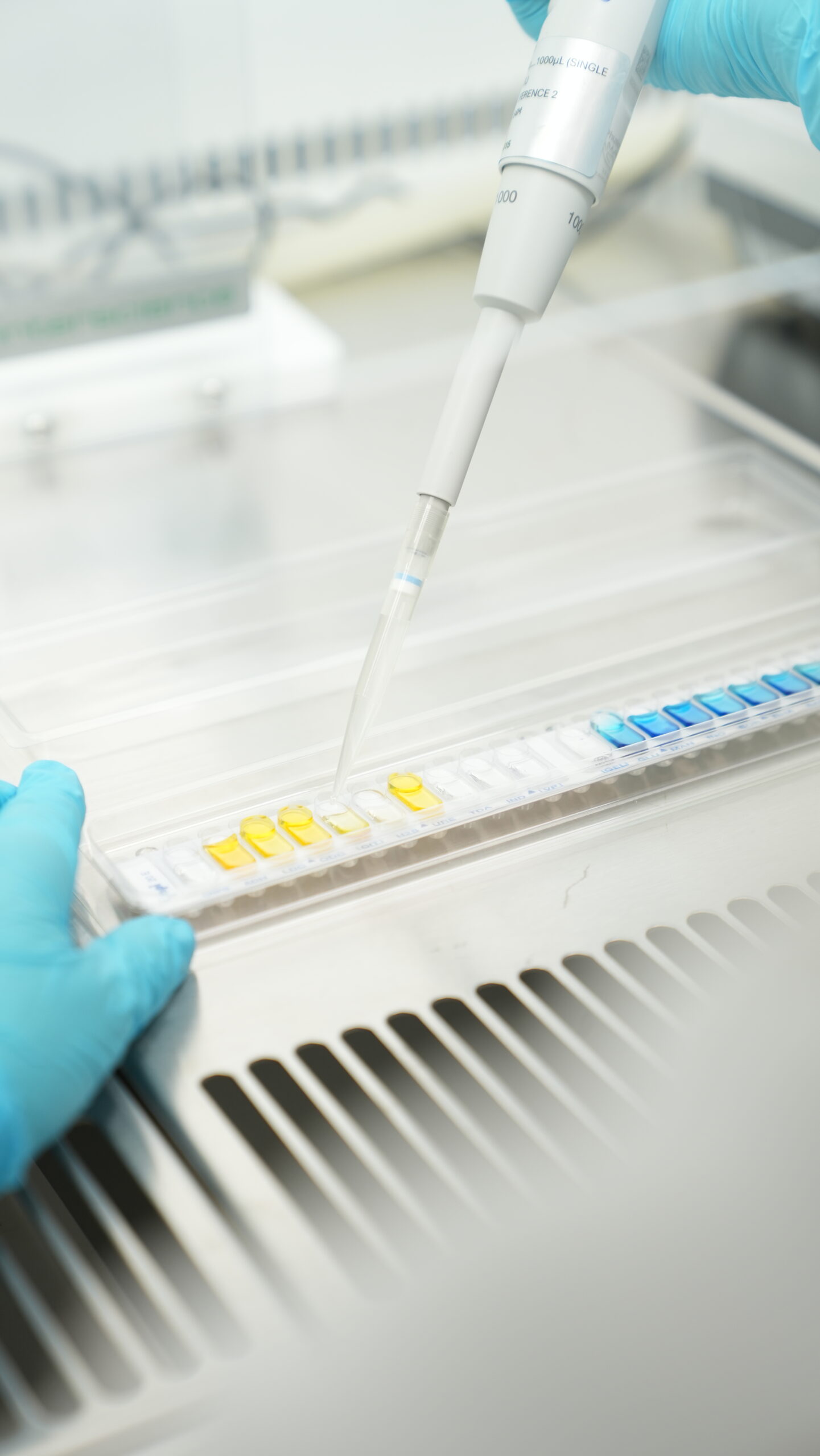

EU-GMP Quality Control Testing
All batches of dried flower are tested in Anthera’s EU-GMP quality control laboratory prior to release. Analytical testing is performed in accordance with the European Pharmacopoeia, including the assessment of stereoscope evaluation, potency, chemical profile, microbiological quality, moisture content, water activity, and other critical quality attributes defined in the product specifications.
Stability studies are conducted under controlled conditions to establish and verify shelf life, in-use stability, and photostability. Final certification and batch release are performed by the certified Qualified Person (QP), ensuring that only products fully compliant with EU-GMP and regulatory requirements are supplied to the market.


Research & Development
Research and Development at Anthera Pharmaceuticals supports the continuous improvement of genetics, cultivation strategies, manufacturing processes, and finished product performance. Cultivars and processes are evaluated for consistency, robustness, and suitability for standardized pharmaceutical production.
Development activities are conducted using structured study designs and data-driven evaluation, enabling optimization of quality attributes, process reliability, and long-term product stability. This approach ensures that Anthera’s dried flower products are not only compliant, but reproducible, scalable, and aligned with pharmaceutical quality expectations.





Cultivation
Plant material intended for extraction is cultivated under Good Agricultural and Collection Practice (GACP) using controlled greenhouse systems and standardized cultivation protocols. Genetics are selected and managed to ensure consistency, traceability, and suitability for pharmaceutical processing.
Cultivation strategies are designed not only to support plant health and yield, but also to ensure chemical reproducibility and predictable composition of the harvested biomass. Environmental parameters such as light, temperature, humidity, irrigation, and nutrition are monitored and adjusted based on cultivar-specific requirements, ensuring that incoming plant material meets predefined quality and processing expectations.


Post-Harvest Processing
Following harvest, plant material enters Anthera’s EU-GMP manufacturing environment and is handled as pharmaceutical starting material. From this stage onward, all processing steps are performed under Good Manufacturing Practice using validated procedures, qualified equipment, and documented workflows.
Post-harvest processing is designed to produce standardized, well-characterized feedstock suitable for extraction. Drying and trimming are performed under defined conditions to achieve controlled moisture content, preserve chemically sensitive constituents, and ensure consistent material quality. Critical quality attributes are assessed to confirm that incoming material meets approved specifications prior to extraction.




Extraction
Extraction at Anthera Pharmaceuticals is designed to enable reproducible pharmaceutical manufacturing while preserving cultivar-specific chemical profiles. Subcritical CO₂ extraction is used to isolate chemically sensitive compounds under controlled temperature and pressure conditions, minimizing degradation of volatile and labile constituents.
The process includes controlled fractionation of terpene, aromatic, and cannabinoid-rich fractions, which are managed as defined intermediates within the manufacturing process. Each fraction is analytically characterized by the EU-GMP quality control laboratory and subsequently used in formulation to produce standardized medicines with defined and reproducible composition.
All extraction and purification steps are performed using validated processes and are fully documented to ensure consistency, traceability, and regulatory compliance.

Packaging
Pharmaceutical extracts and finished formulations are packaged under EU-GMP conditions using controlled environments and validated filling and closing processes. Packaging materials are selected to protect the product from light, oxygen, moisture, and external contamination and to maintain stability throughout the product’s shelf life.
All packaging operations are subject to in-process controls, reconciliation, and full batch documentation. Each packaged batch is verified against approved specifications before release, ensuring product integrity, patient safety, and full regulatory compliance.

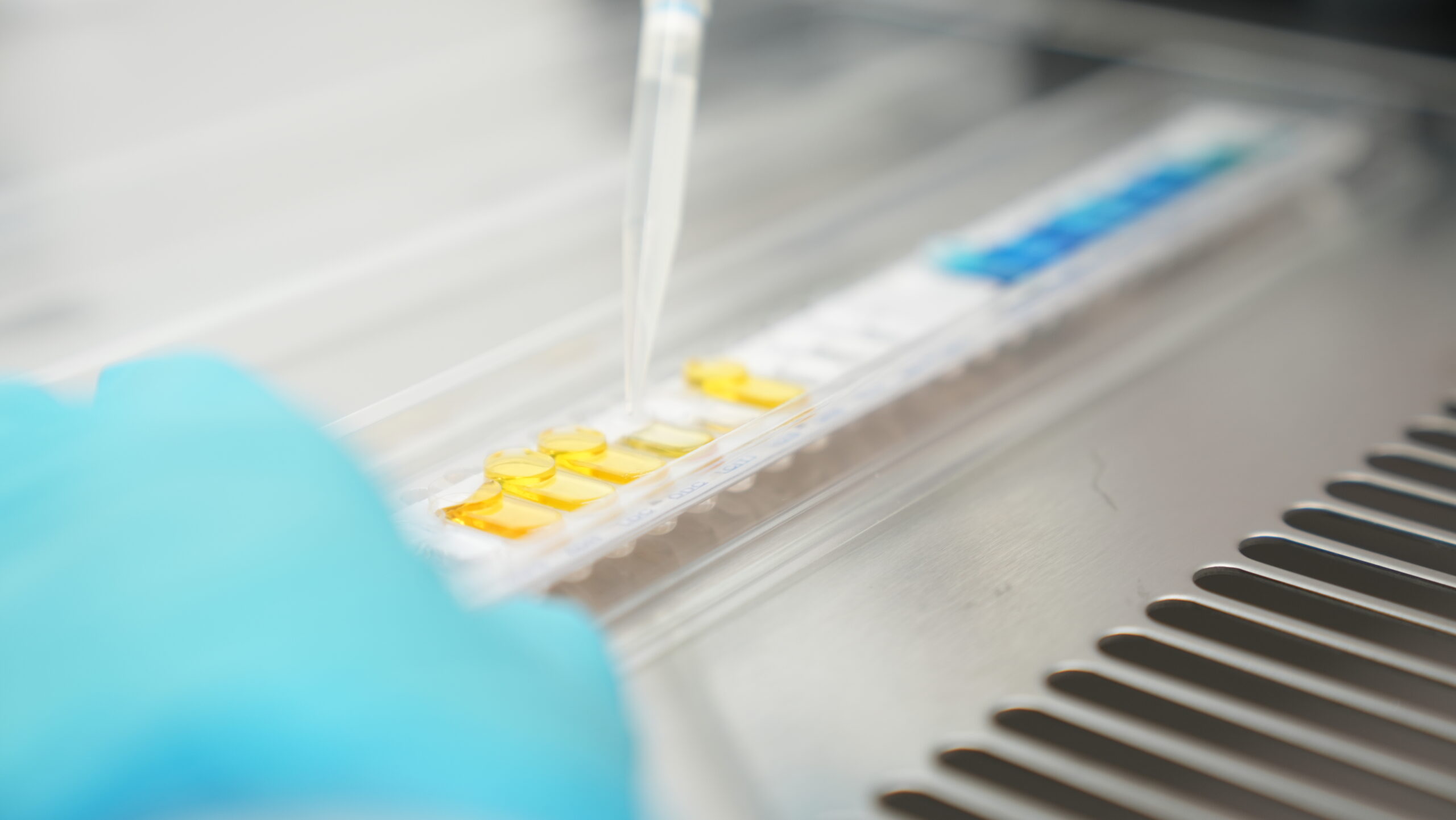



EU-GMP Quality Control testing
All extracted intermediates and finished formulations are tested in Anthera’s EU-GMP quality control laboratory prior to release. Testing is performed according to approved analytical methods and includes assessment of identity, potency, chemical profile, impurities, and other critical quality attributes defined in the product specifications.
Analytical activities are conducted in accordance with the European Pharmacopoeia and applicable requirements of the European Medicines Agency (EMA) and the National Organization for Medicines in Greece (EOF). Final batch certification and release are performed by the Qualified Person (QP), ensuring that only compliant products are supplied to the market.



Research & Development
Research and Development at Anthera Pharmaceuticals supports the ongoing optimization of genetics, extraction processes, and finished formulations. Cultivars and formulations are evaluated based on chemical profile, process robustness, stability, and suitability for standardized pharmaceutical production.
Development programs follow structured, data-driven methodologies, with iterative cycles of testing and optimization used to support process validation, quality consistency, and long-term product reliability. This approach enables the development of a well-characterized, scalable portfolio of medical cannabis medicines aligned with pharmaceutical quality standards and regulatory expectations.



